BPC-157 is everywhere right now. Forums, fitness influencers, biohacking podcasts — everyone seems to have a strong opinion about a peptide most physicians have never prescribed, and regulators have never approved. Search volume for this compound has surged even after the FDA restricted it in 2023, which tells you something about how much demand outpaces the available evidence.
If you’ve been researching BPC-157, you’ve probably already noticed that the enthusiasm and the science don’t quite match up. Advocates describe it as a breakthrough healing compound. Critics call it unproven hype. The honest answer sits somewhere more complicated than either camp wants to admit.
Here’s what the research actually shows — and what anyone seriously considering this compound should understand before going further.
What BPC-157 Is
BPC-157 stands for Body Protection Compound-157. It’s a synthetic pentadecapeptide — a chain of 15 amino acids — originally isolated from a protein found in human gastric juice. Researchers at the University of Zagreb first described it in 1992, and the bulk of the published science has come from that same Croatian laboratory ever since.
That last sentence matters more than most people realize.
The peptide has one quality that distinguishes it from most short-chain compounds: stability. The majority of peptides are broken down rapidly by enzymes in the gut, bloodstream, or peripheral tissues. BPC-157 resists that enzymatic degradation unusually well, which is why it can be administered orally in animal models without being destroyed before it reaches target tissues. That stability is also what attracted researchers to it in the first place, and why it remains one of the more studied investigational peptides in preclinical literature.
How It Works — What Preclinical Studies Suggest
The mechanistic picture is the strongest part of BPC-157’s scientific story, even though that story comes almost entirely from laboratory and animal research.
In preclinical studies, BPC-157 has been reported to interact with signaling pathways, including VEGFR2-Akt-eNOS and FAK-paxillin, that are involved in tissue repair processes. These findings come from cell culture and animal models — they do not demonstrate clinical benefit in humans. With that context firmly in place, the preclinical mechanisms are worth understanding because they explain why researchers have found the compound worth studying.
The VEGFR2-mediated pathway, when activated in rodent models, appears to increase nitric oxide production and promote vasodilation. The FAK-paxillin activation supports cell migration and adhesion. JAK2/STAT signaling, also observed in these models, relates to cell survival. The peptide has also shown upregulation of Egr-1, a gene associated with angiogenesis, and suppression of NF-κB, a central mediator of inflammatory signaling. Heat shock protein induction — specifically HSP70 and HSP90 — has been documented in vitro as well [1].
This is a mechanistically plausible profile for a tissue repair compound. The problem, and it’s not a small one, is that mechanistic plausibility in rodents has a notoriously poor track record of translating to clinical outcomes in people.
What the Research Actually Shows
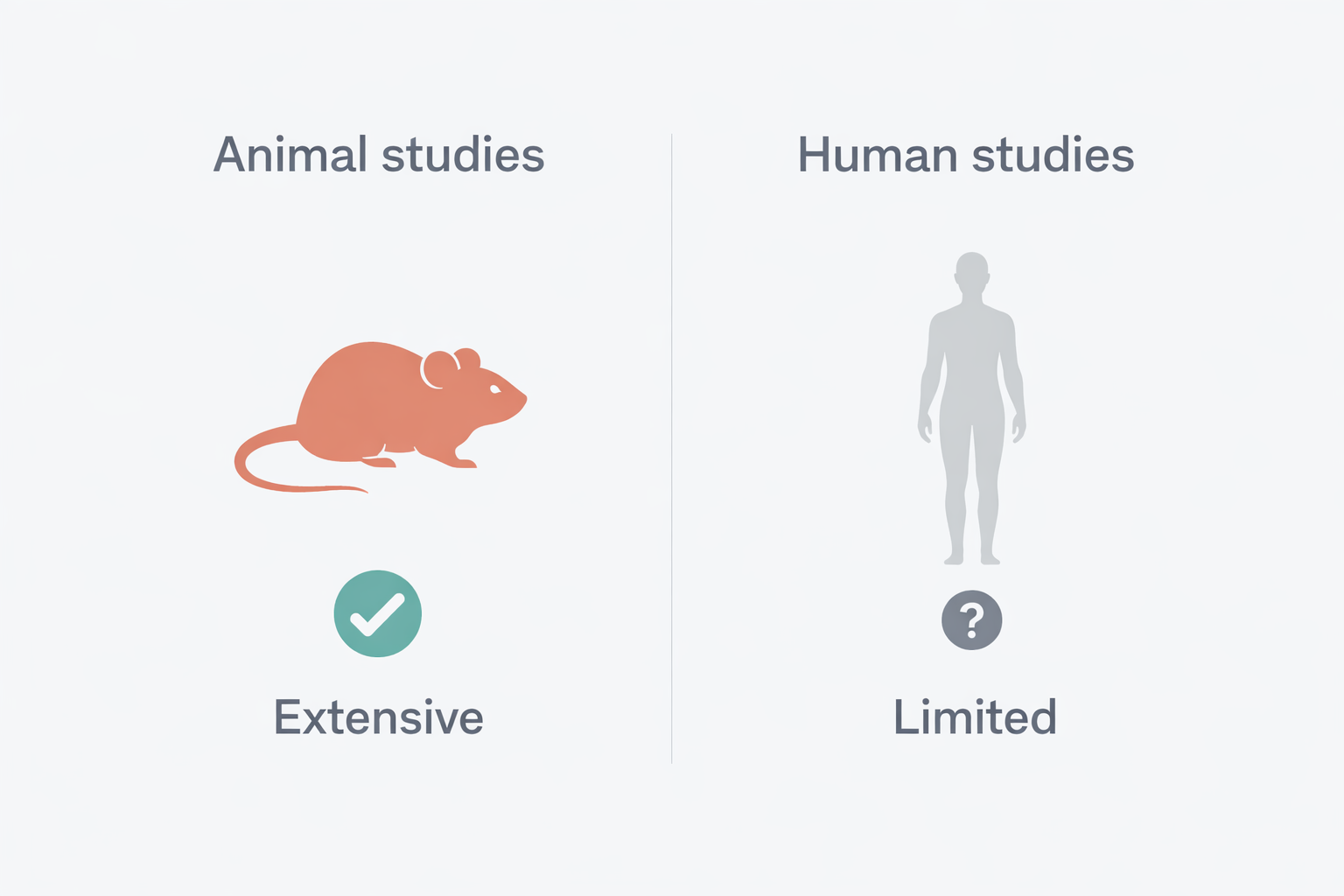
This is where most online coverage of BPC-157 loses the plot entirely.
Preclinical data: Extensive. Dozens of published rodent studies report improved tissue repair markers and biomechanical outcomes in tendon injury models through ERK1/2-related pathways [2], better functional recovery after muscle crush injuries [3], resolution of experimentally induced gut lesions and vascular disturbances [4], and wound healing activity via VEGFR2-mediated angiogenesis [5]. The volume is genuinely unusual for an unapproved compound — most investigational peptides have far thinner preclinical files.
Human data: Essentially absent in terms of efficacy.
The most significant human study published to date is a 2025 pilot safety study by Lee and Burgess [6]. Two participants received intravenous BPC-157 at doses up to 20 mg. No adverse effects were observed on vital signs, blood biomarkers, or subjective symptoms. This is a meaningful safety signal — it suggests the compound may be tolerated at that dose in healthy adults. But two participants, no control group, no efficacy endpoints: this is not a treatment study. It tells us almost nothing about whether the compound does anything useful in humans.
One active registered clinical trial (NCT07437547, Hudson Biotech, 2026) is currently recruiting approximately 120 participants to evaluate subcutaneous BPC-157 versus placebo for MRI-confirmed healing in grade II hamstring strains [7]. Results are not expected before 2027.
That is the complete human evidence base as of March 2026.
The gap between the animal literature and human data is not a technicality. It’s the central clinical problem. Compounds that perform impressively in rodent injury models frequently fail to translate when tested in people — this is one of the most consistent patterns in drug development. We simply do not know yet whether BPC-157 is one of those failures or something different.
The Gastrointestinal Research
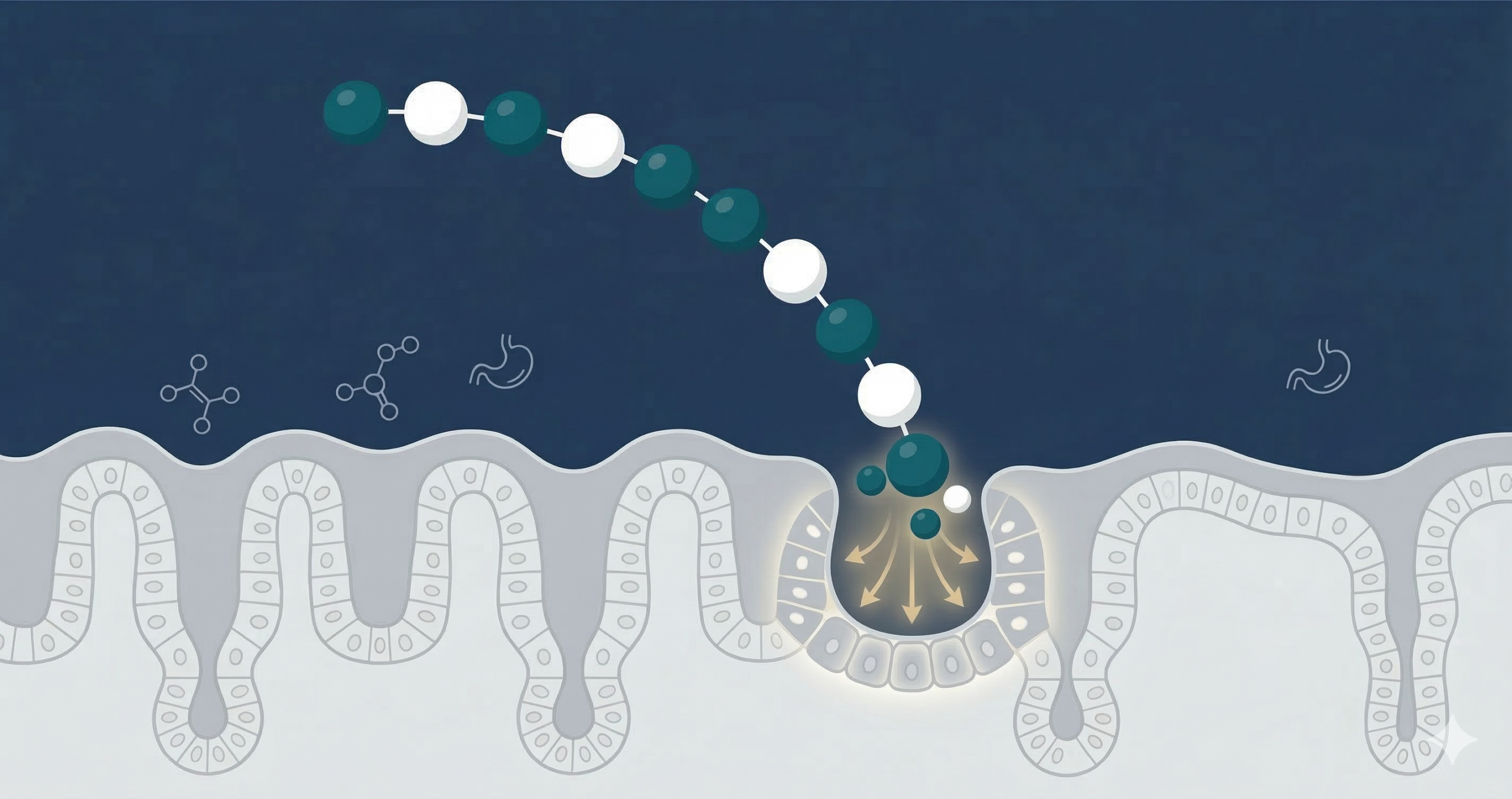
Because the peptide was originally identified for its gastroprotective properties, the gastrointestinal preclinical data deserves its own section.
In animal gastrointestinal models, BPC-157 has demonstrated mucosal protective effects, including reversal of experimentally induced NSAID damage and inflammatory bowel-like lesions [4]. The oral stability of the compound is relevant here — unlike most peptides that are degraded before reaching the intestine, BPC-157 survives the acidic gastric environment, making oral delivery at least theoretically feasible. It is worth noting, however, that stability in the gut and systemic bioavailability are two different things. How much of an orally administered dose actually enters systemic circulation — versus remaining localized to the gut lumen — is still largely unknown in humans. This distinction matters practically: someone taking oral BPC-157 hoping for tendon or muscle repair elsewhere in the body should not assume that gut survival translates to meaningful systemic exposure.
Because the peptide demonstrated these mucosal protective effects in animal gastrointestinal models, researchers have explored its potential relevance to digestive system physiology. However, there are currently no human clinical trials demonstrating safety or efficacy for any gastrointestinal condition. Any discussion of BPC-157 and gut health remains strictly in the domain of preclinical hypothesis.
FDA Regulatory Status in 2026
This is the area where patients most often receive inaccurate information, sometimes from sources that should know better.
In September 2023, the FDA placed BPC-157 on its Category 2 bulk substances list under Sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act. Category 2 classification means the agency has identified potential significant safety risks — citing immunogenicity concerns, risk of peptide-related impurities, and insufficient human safety data for the proposed routes of administration [8].
The practical consequence: licensed 503A compounding pharmacies cannot legally compound injectable BPC-157 for human use. The FDA has issued warning letters to pharmacies that continued compounding after the classification took effect.
Unlike CJC-1295 and ipamorelin — which were temporarily removed from the Category 2 list in late 2024 pending Pharmacy Compounding Advisory Committee review — BPC-157’s injectable form remains classified as a substance with significant safety concerns as of this writing. Oral and topical forms occupy a somewhat different regulatory position, though they remain unapproved drugs without any approved indication.
The regulatory landscape may be shifting. Current HHS leadership has publicly challenged several of the 2023 Category 2 classifications, and a broader reconsideration of the compounding restrictions is under active discussion. A final rule on related peptide compounds is expected no later than March 2027. But as of today, injectable BPC-157 from a licensed compounding pharmacy is not a legal option in the United States.
Patients should verify the regulatory status of any peptide product with their physician and pharmacy before considering its use.
Who Should Approach This Compound With Caution
Because human safety data is limited, certain patient populations carry meaningful theoretical risk based on the compound’s preclinical mechanistic profile. These are not arbitrary cautions — each one follows from what the animal and cell data actually suggest:
Cancer history or active malignancy. The compound has been shown in preclinical models to promote angiogenesis and cell proliferation. In injured tissue, these are repair processes. In a patient with a cancer history, the same signaling could theoretically support tumor vascularization. No human data exists to quantify this risk, which is precisely why it deserves serious attention.
Pregnancy and lactation. No safety data in any human population. The preclinical reproductive data are insufficient to draw any conclusions.
Autoimmune disease and immunosuppressed patients. The compound modulates immune signaling pathways. In someone already on immunosuppressive therapy or with active autoimmune pathology, the direction and magnitude of that modulation is unpredictable.
Pediatric and elderly populations. The pharmacokinetic data that exists comes from animal studies. Extrapolating dosing or safety parameters across age extremes is not supportable with current evidence.
Because human clinical trials remain limited, the full safety profile of BPC-157 is simply unknown. Potential risks could include immune reactions, peptide-related impurities, or unforeseen biological effects not captured in animal models. Anyone considering this compound should operate under the assumption that unknown risks exist until larger controlled human trials are completed.
How BPC-157 Compares to Related Compounds
For context, it helps to understand where BPC-157 sits relative to other peptides that come up in similar discussions.
TB-500, a fragment of thymosin beta-4, is frequently mentioned alongside BPC-157 in the research literature. The two have been examined in separate animal models for complementary repair-related mechanisms — BPC-157 with stronger local tissue effects in preclinical models, TB-500 with more emphasis on actin regulation and systemic distribution. Online forums and non-regulated sources often discuss combining these compounds, although there are no controlled human trials evaluating such combinations, and both remain in similar regulatory positions.
Other peptides discussed in longevity and hormone optimization contexts — such as CJC-1295 and ipamorelin — fall under separate regulatory discussions regarding compounding eligibility and have distinct mechanisms unrelated to tissue repair.
For patients with specific concerns about gastrointestinal health, musculoskeletal recovery, or growth hormone optimization, these are meaningfully different conversations that deserve individualized medical evaluation.
Where the Evidence Actually Stands
The mechanistic science behind BPC-157 is scientifically interesting but remains preliminary. The animal evidence is extensive by the standards of investigational peptides. The 2025 pilot safety data in two human participants is, at a minimum, not alarming. I understand why patients with persistent orthopedic issues or treatment-resistant gut problems find themselves researching this compound.
But there are no completed randomized controlled trials demonstrating that BPC-157 improves any clinical outcome in human beings. Every claim of efficacy — in tendons, muscles, or gastrointestinal tissue — is extrapolated from rodent and in vitro data. That gap matters enormously.
From a clinical perspective, the most important study currently underway is the randomized trial (NCT07437547) expected to report in 2027. Its results will tell us more than a decade of accumulated anecdotal reports. The pending regulatory decisions from the FDA and PCAC will also reshape what’s legally available to patients in ways that may look quite different in 12 months than they do today.
At Androgenix, our approach to hormone optimization and peptide science is grounded in what the evidence currently supports and what the law currently permits. We track regulatory developments closely and adjust our clinical approach accordingly.
If you want a clear picture of where peptide science fits within a comprehensive hormone optimization strategy — based on your specific labs, history, and goals — that conversation starts with a proper evaluation.
References
- Vukojević J et al. (2025). BPC-157 mechanisms in tissue repair. Front Pharmacol. PMC12446177.
- Gwyer D et al. (2019). BPC-157 and wound healing via VEGFR2. Cell Tissue Res (review).
- Sikiric P et al. (2018). Muscle recovery and BPC-157. Curr Pharm Des.
- Seiwerth S et al. (2018). Organ protection and thrombosis models. Curr Pharm Des.
- Tohyama H et al. (2022). Pharmacokinetics of BPC-157. Front Pharmacol.
- Lee E, Burgess K. (2025). Intravenous BPC-157 pilot safety study, n=2. Altern Ther Health Med. PMID: 40131143.
- Hudson Biotech. NCT07437547. Phase 2 RCT: BPC-157 for hamstring strain. ClinicalTrials.gov. 2026.
- U.S. Food & Drug Administration. Certain Bulk Drug Substances for Use in Compounding that May Present Significant Safety Risks. Updated 2025. fda.gov.
BPC-157 is not approved by the U.S. Food and Drug Administration for the diagnosis, treatment, or prevention of any disease. This article is strictly educational and does not imply clinical availability. Because human clinical trials are limited, the safety profile of BPC-157 remains uncertain — potential risks include immune reactions, peptide impurities, and unforeseen biological effects. Patients should not attempt to obtain peptides from unregulated or research chemical suppliers. Consult a licensed physician before making any decisions related to peptide compounds.